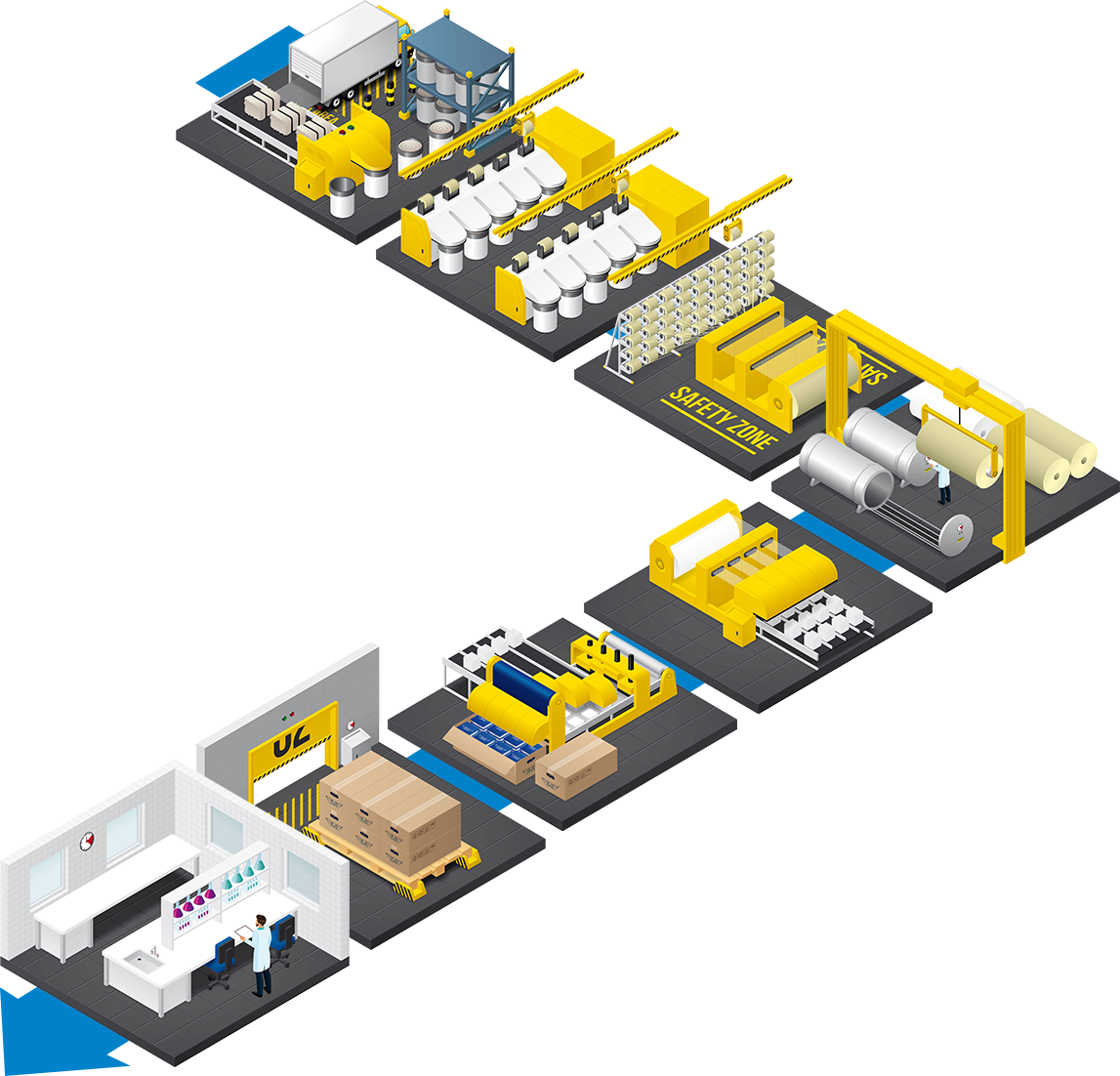
Da rama de algodão à expedição do produto final, é percorrido um longo caminho. O recurso às melhores matérias-primas na empresa ADA FIOS, único fabricante europeu de tecido gaze, e à tecnologia mais avançada desenvolvida pela empresa ADATEC, permite à empresa ADA concentrar todo o seu potencial na produção de compressas, com qualidade reconhecida e certificada por competentes organismos externos.



